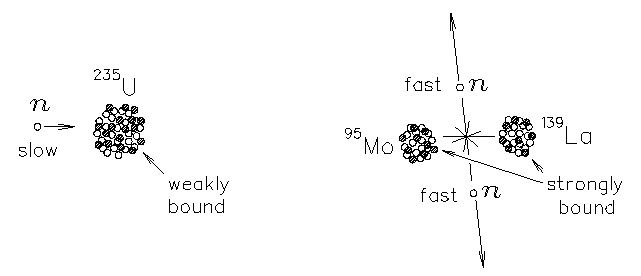
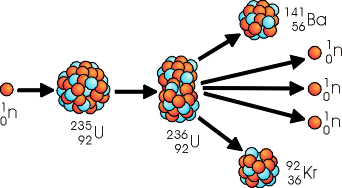
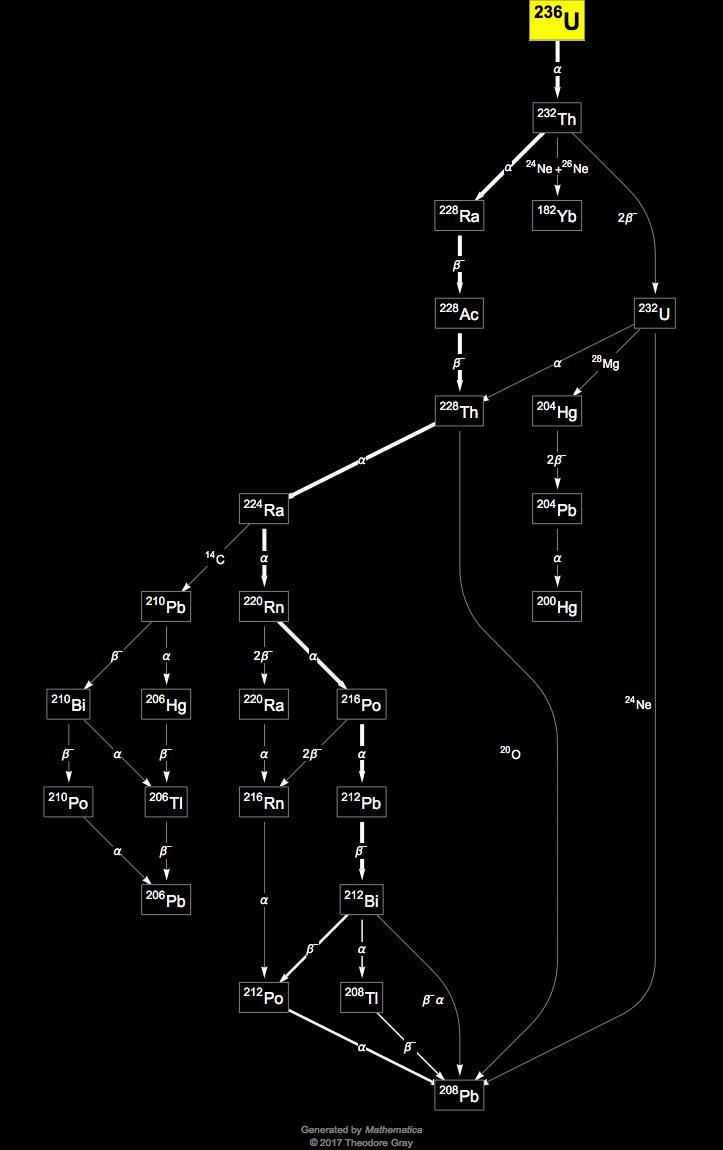
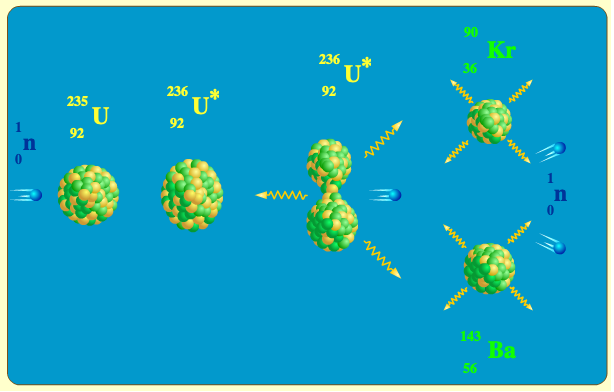
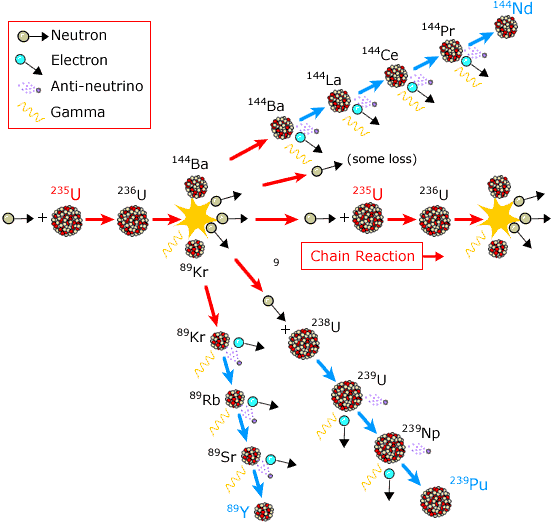
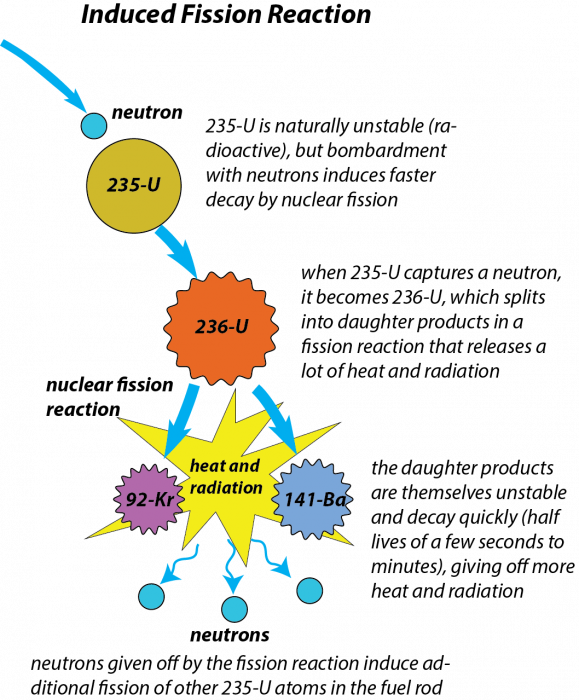
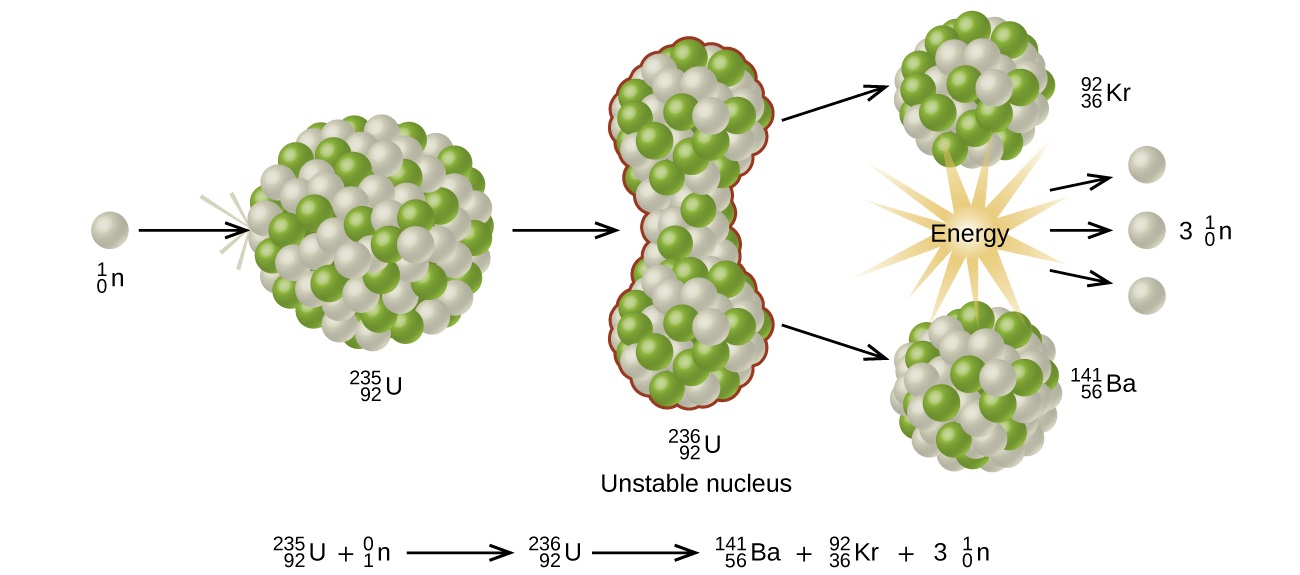
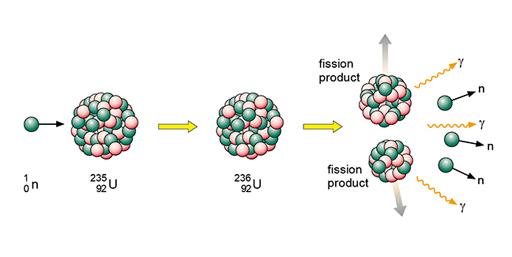
If mass of U^(235)=235.12142 a.m.u., mass of U^(236) =236.1205 a.m.u, and mass of neutron =1.008665 a.m.u, then the energy required to remove one neutron from the nucleus of U^(236) is nearly about.

Temporal change of 236U/238U and 235U/238U isotopic ratios in atmospheric deposition in Tokyo and Akita from 1963 to 1979 - ScienceDirect
The fission cross sections of /sup 230/Th, /sup 232/Th, /sup 233/U, /sup 234/U, /sup 236/U, /sup 238/U, /sup 237/Np, /sup 239/Pu and /sup 242/Pu relative /sup 235/U at 14. 74 MeV neutron
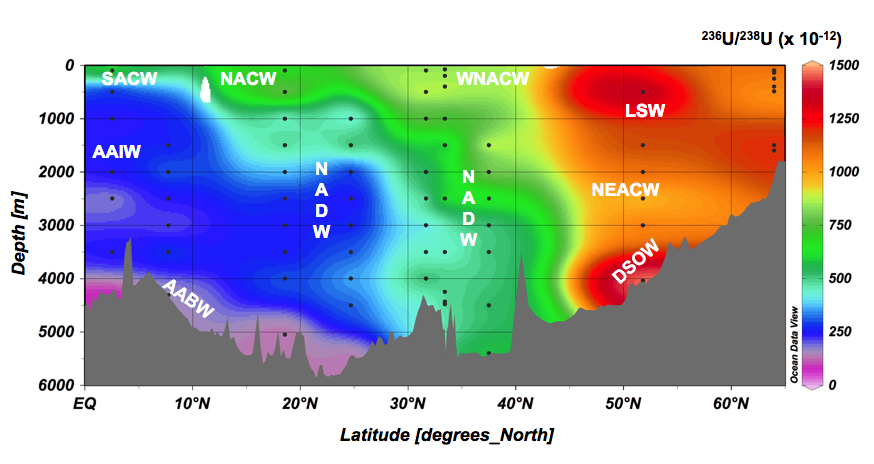
The potential of anthropogenic 236-Uranium as a new and transient oceanographic tracer demonstrated in the North Atlantic – GEOTRACES

SOLVED: An excited Uranium (U) nucleus decays at rest into Rb, Cs, and several neutrons 236 U 92 143 Cs 55 90 Rb 37 In terms of atomic mass units ( u) the
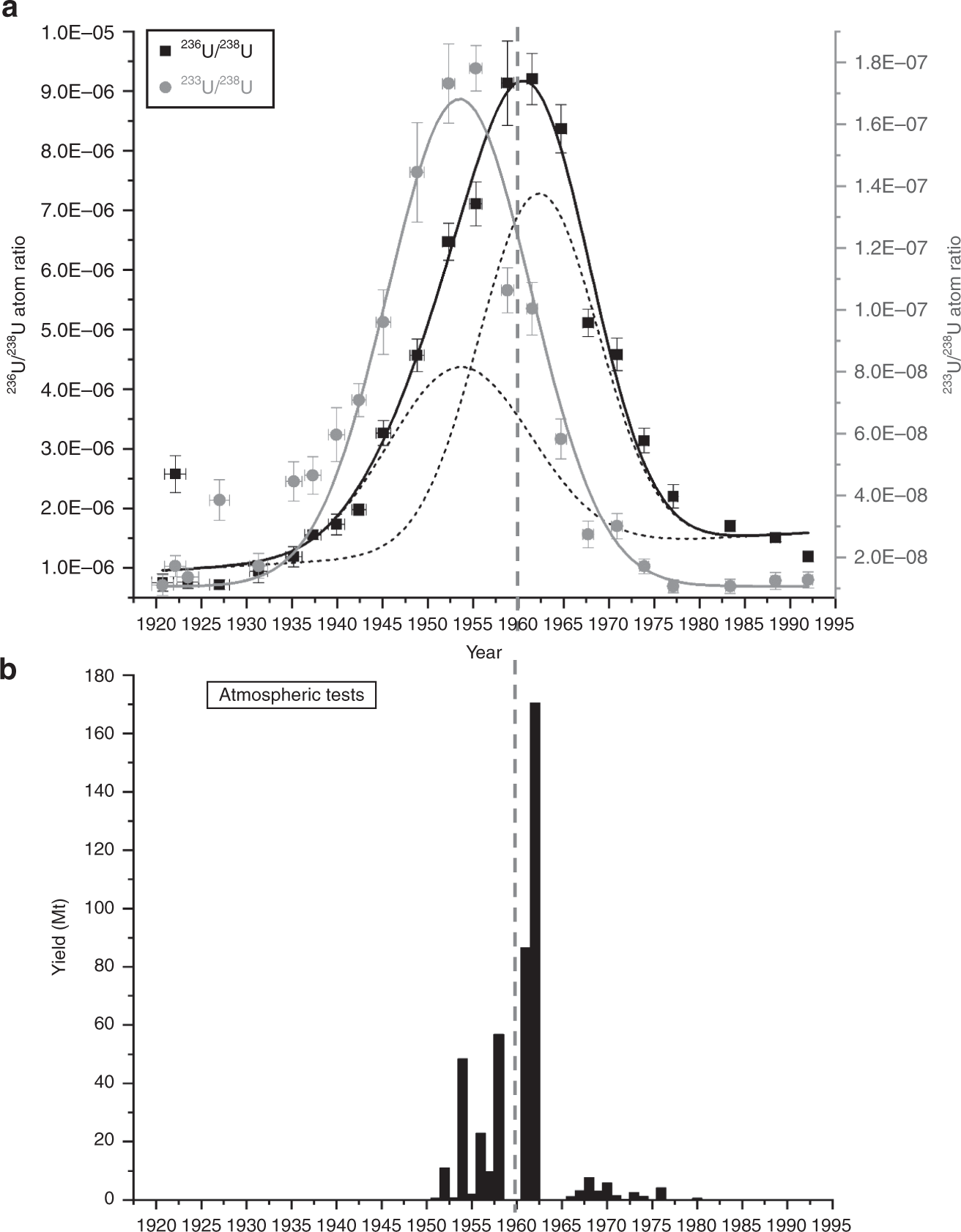
233U/236U signature allows to distinguish environmental emissions of civil nuclear industry from weapons fallout | Nature Communications