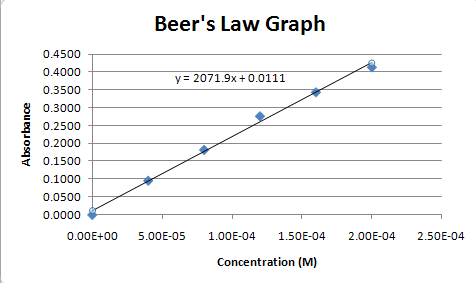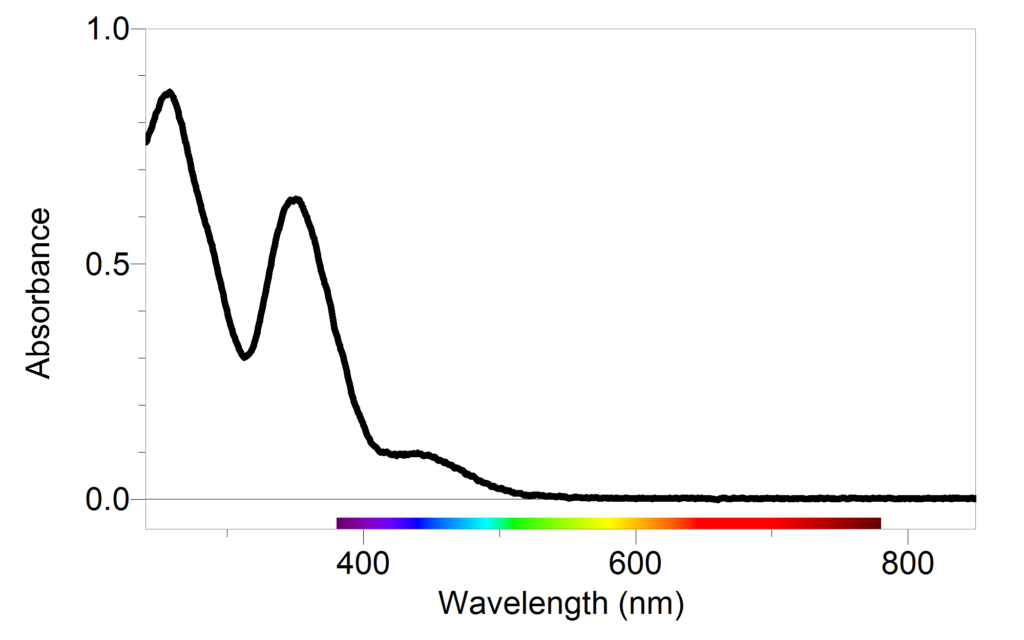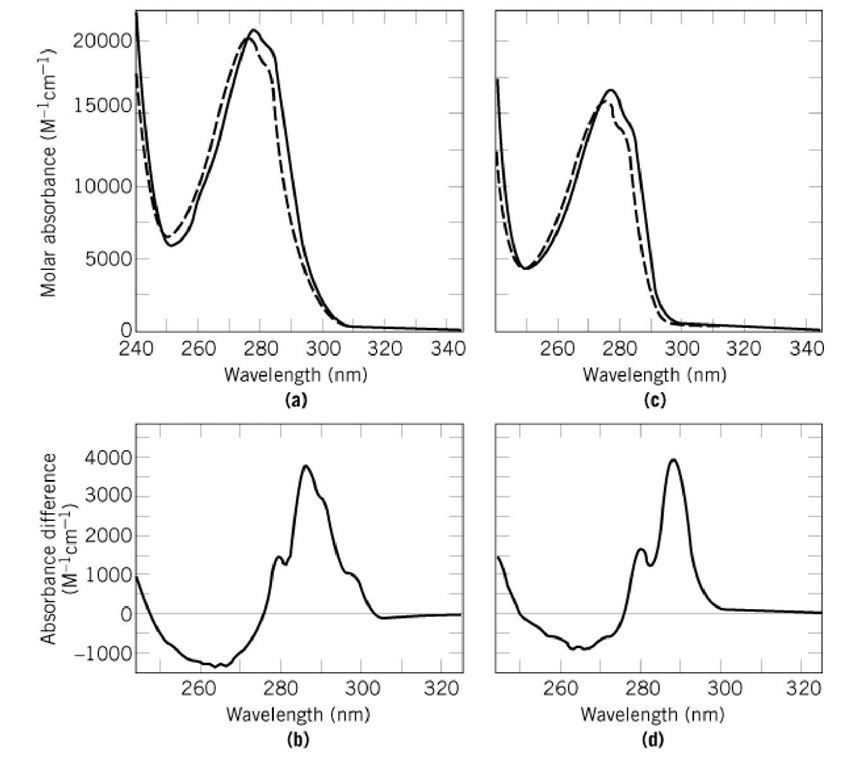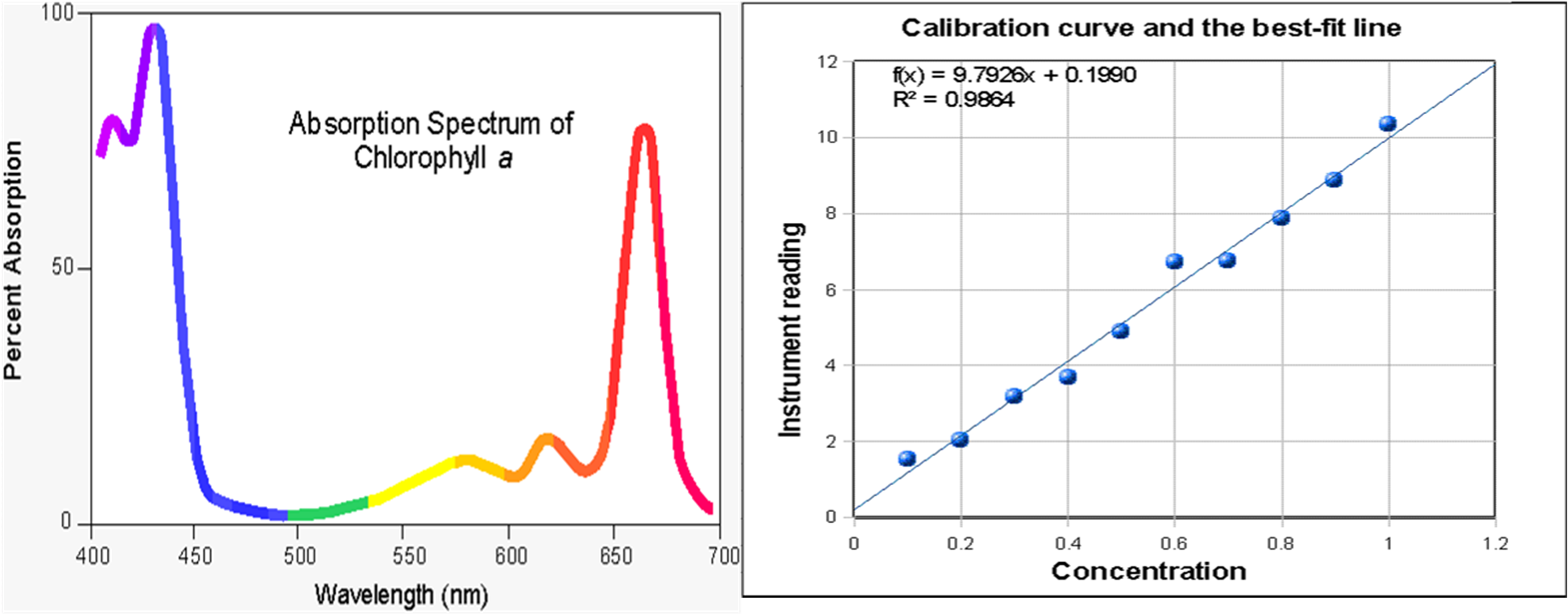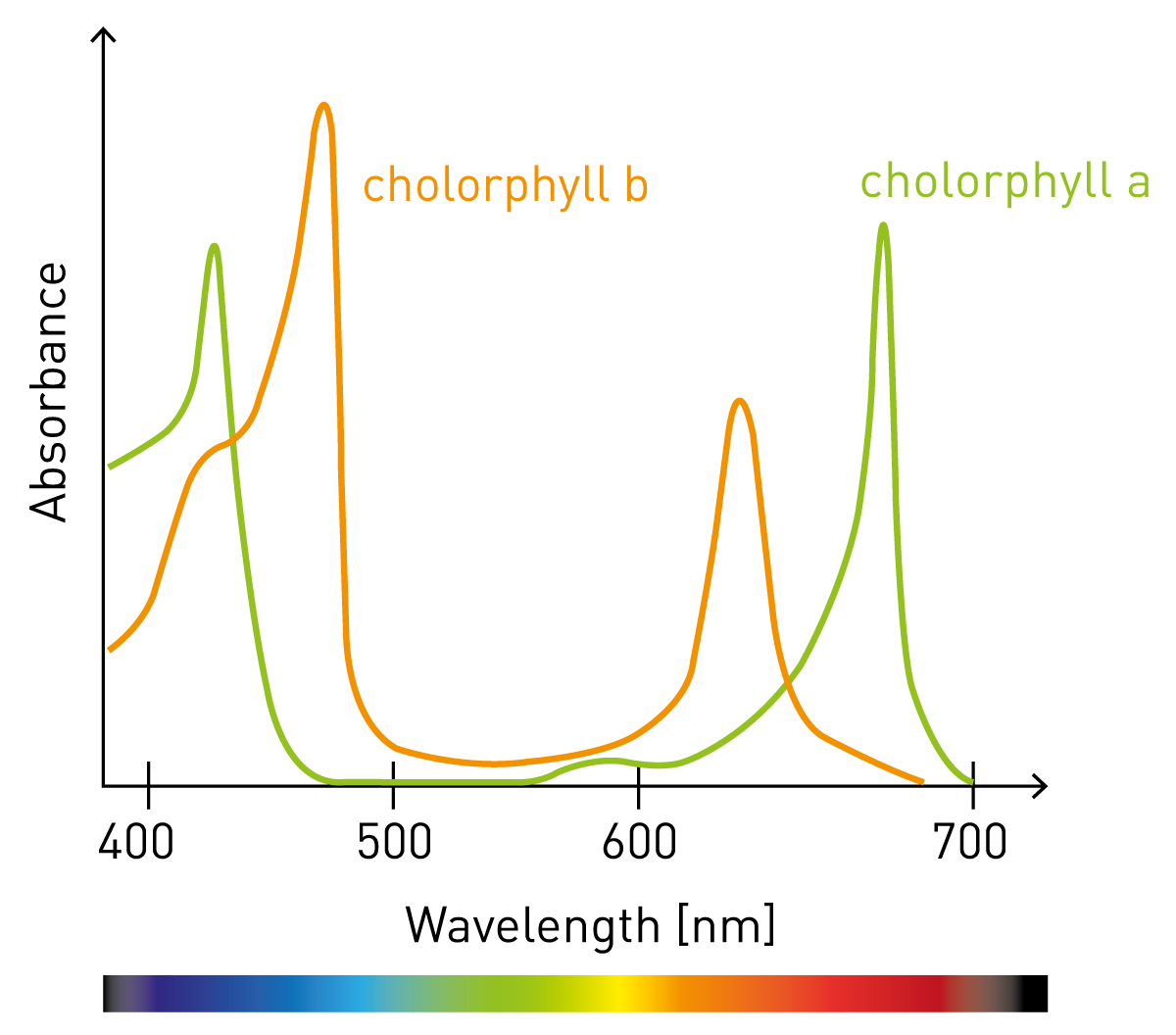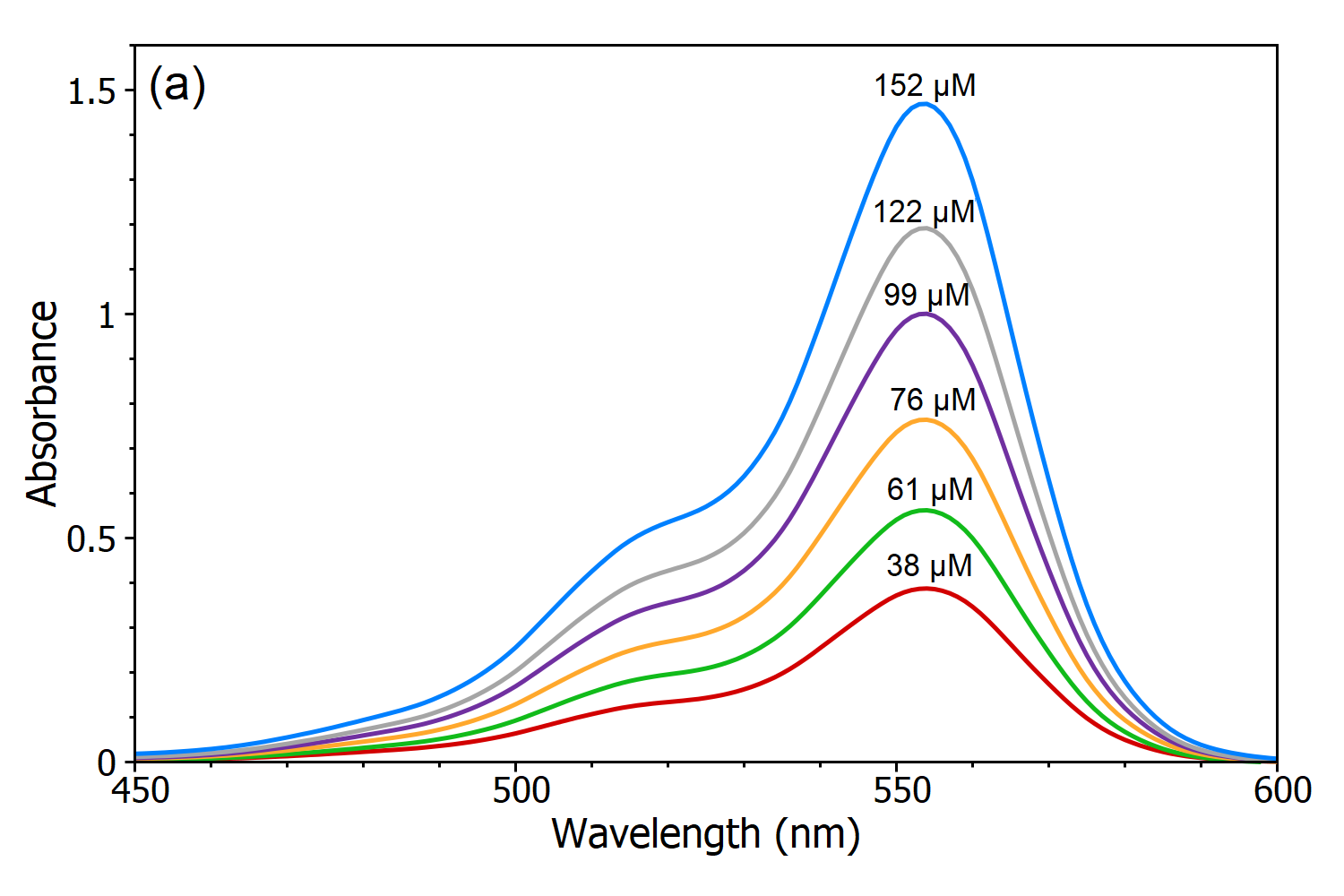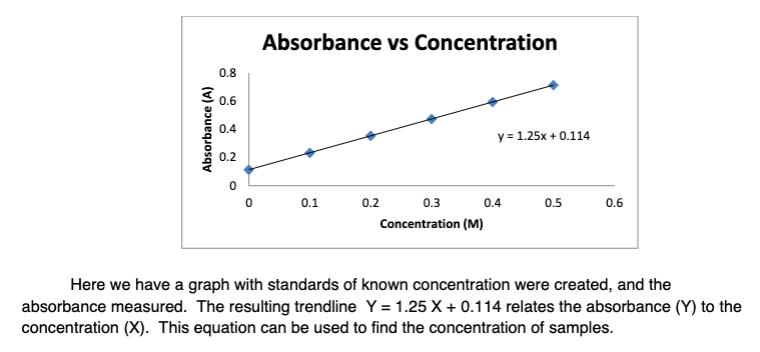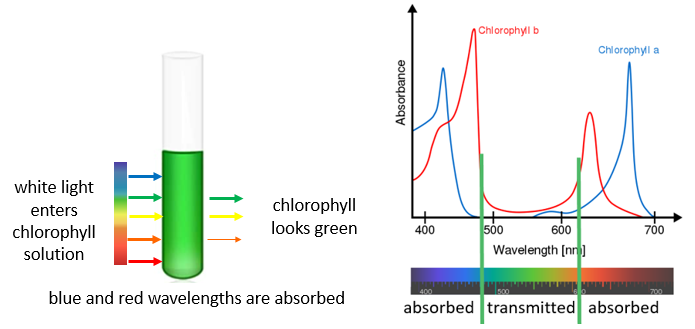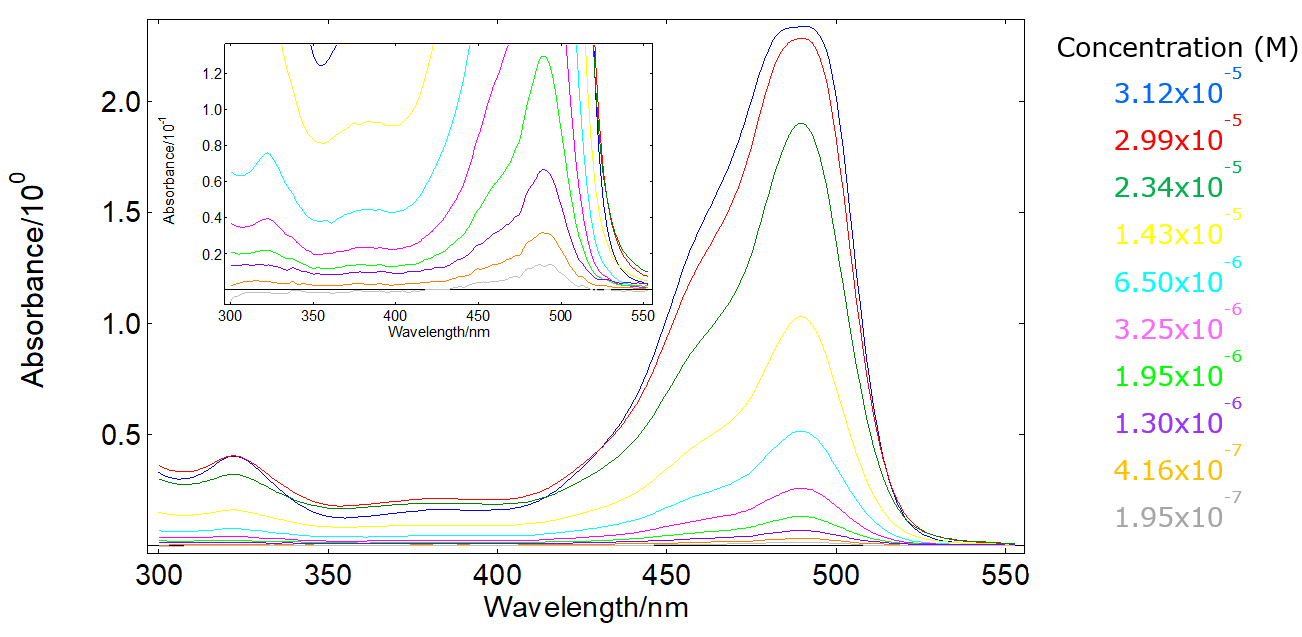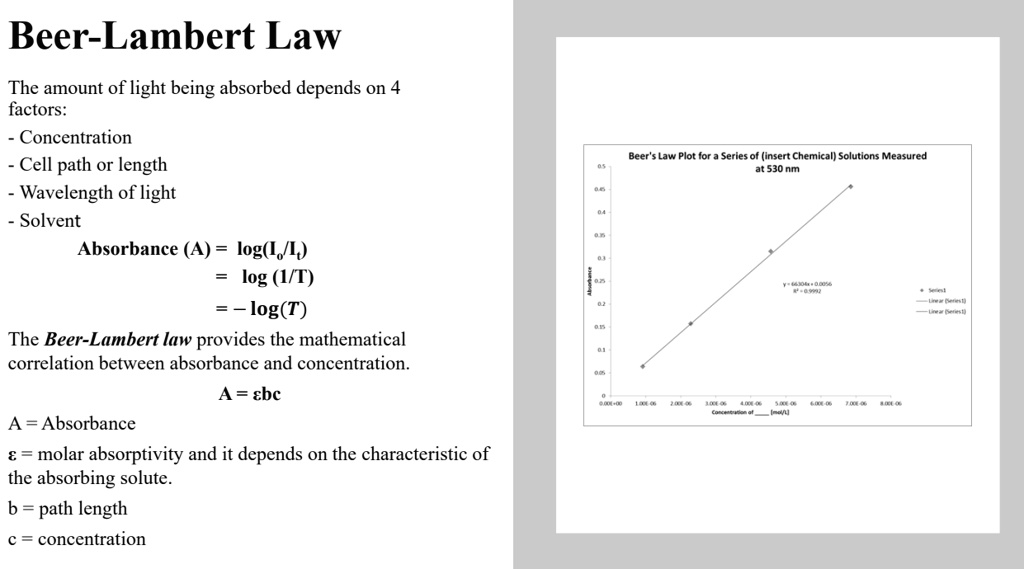
SOLVED: Beer-Lambert Law The amount of light being absorbed depends on 4 factors: Concentration Cell path or length Wavelength of light Solvent Absorbance (A) = log(LJI) log (IT) log(T) The Beer-Lambert law

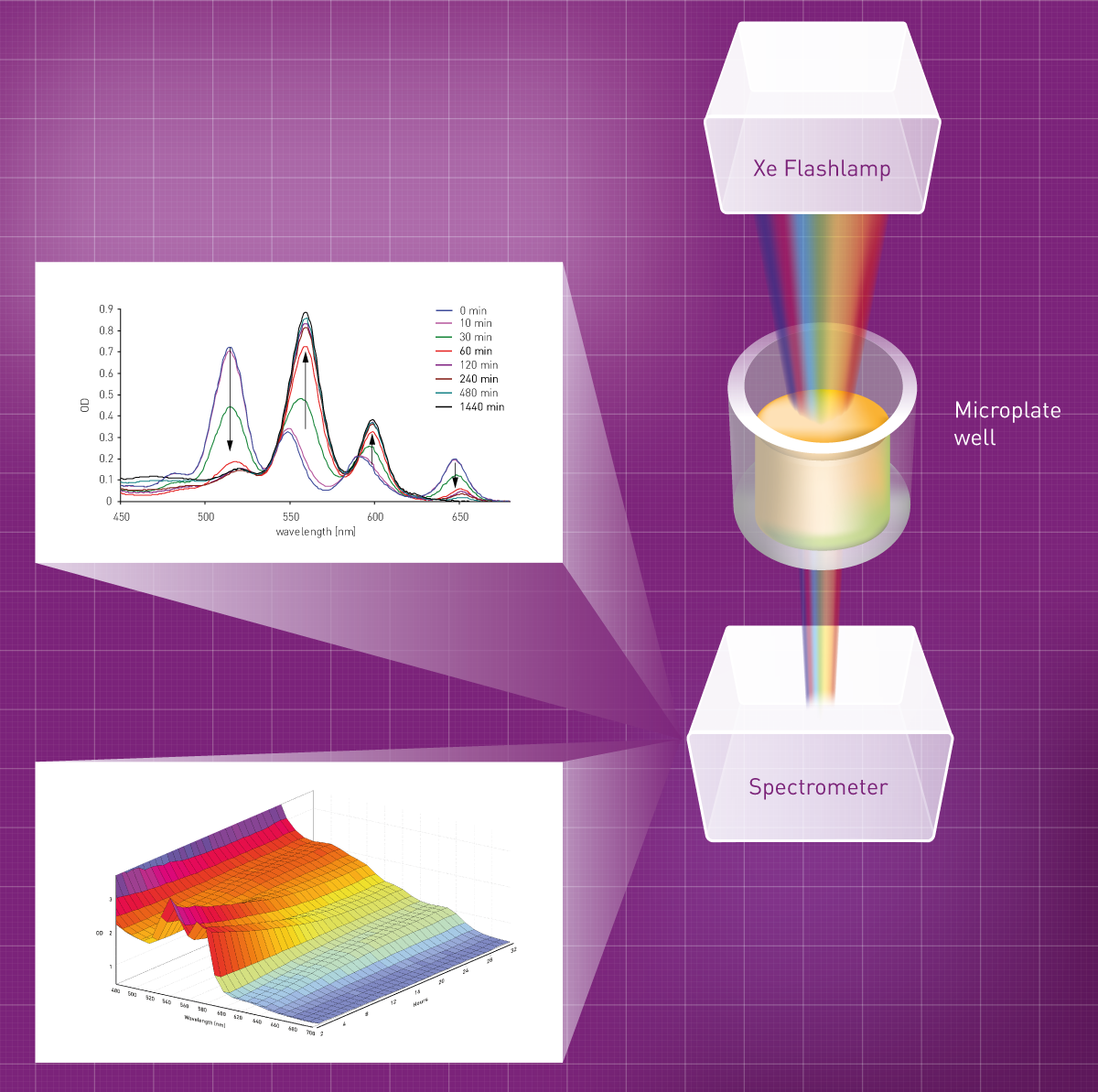
Absorbance Measurements – the Quick Way to Determine Sample Concentration - Eppendorf Handling Solutions

Absorbance Measurements – the Quick Way to Determine Sample Concentration - Eppendorf Handling Solutions