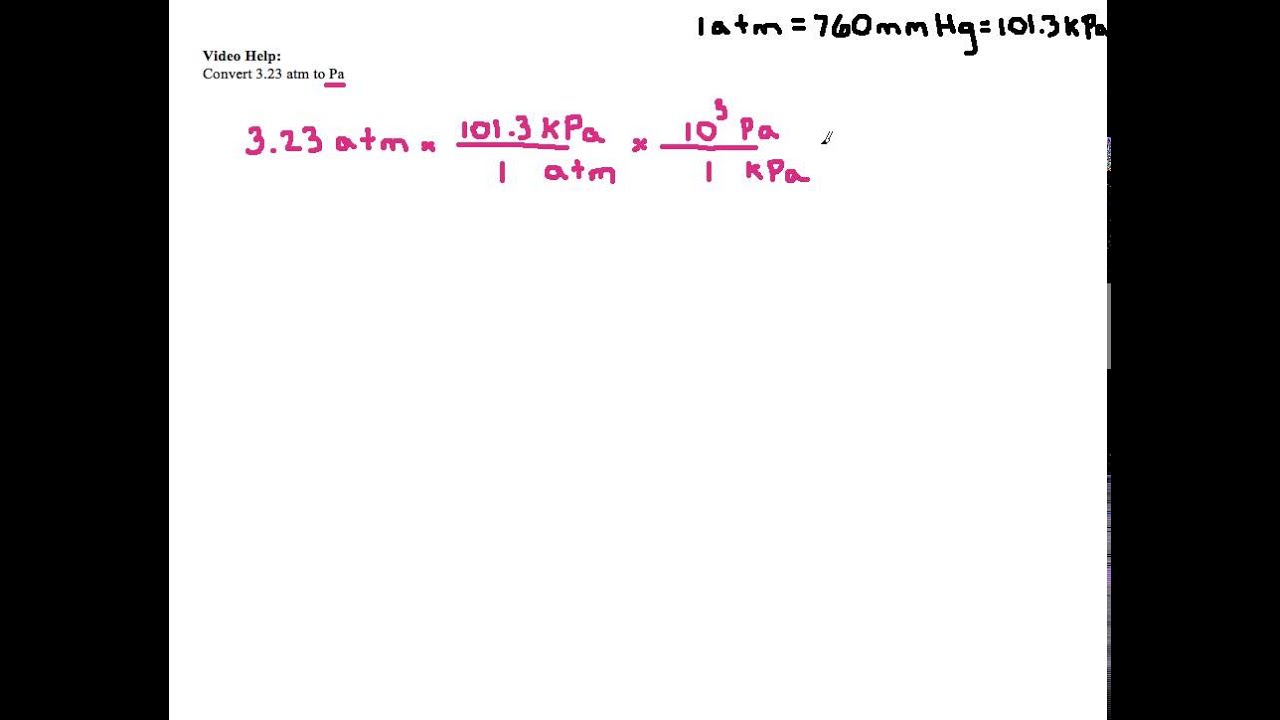24-h ICP (mmHg) and PA (mmHg) in IIH patients with PT (n = 5) and NPT... | Download Scientific Diagram

homework and exercises - How is formula for converting pressure from mmHg to Pa derived? - Physics Stack Exchange

SOLVED: Convert 661,000 pascal, pa into torr: 1atm = 760 torr = 760 mmHg = 101,325 pa 101.325 kpa 1.01325 barr 14.696 p.s.i. 29.921 inHg (inHg and p.s.i. are approximations of the metric pressures and are subject to significant figures)

SOLVED: A student reads a barometer in the laboratory and finds the prevailing atmospheric pressure to be 709 mm Hg. Express this pressure in atmospheres, kilopascals, torrs, pounds per square inch, and
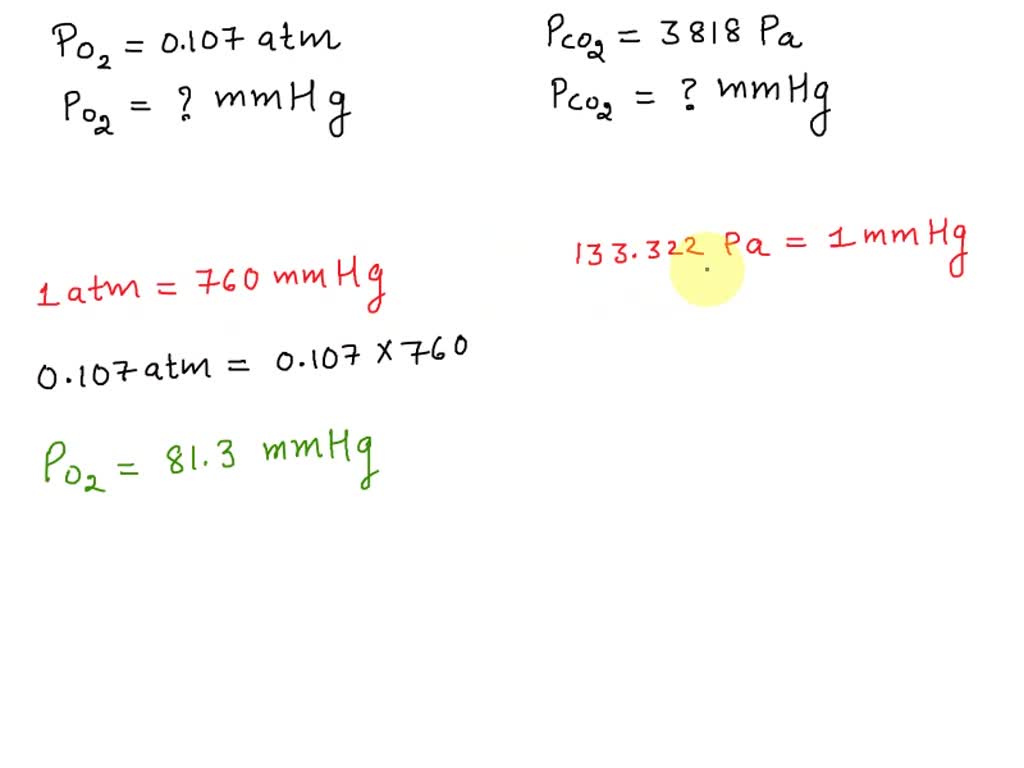
SOLVED: The patient's partial pressure of O2 is at 0.107 atm. What is the partial pressure in mmHg? Pa O2= mmHg The partial pressure of CO2 is 3818 Pa. What is the

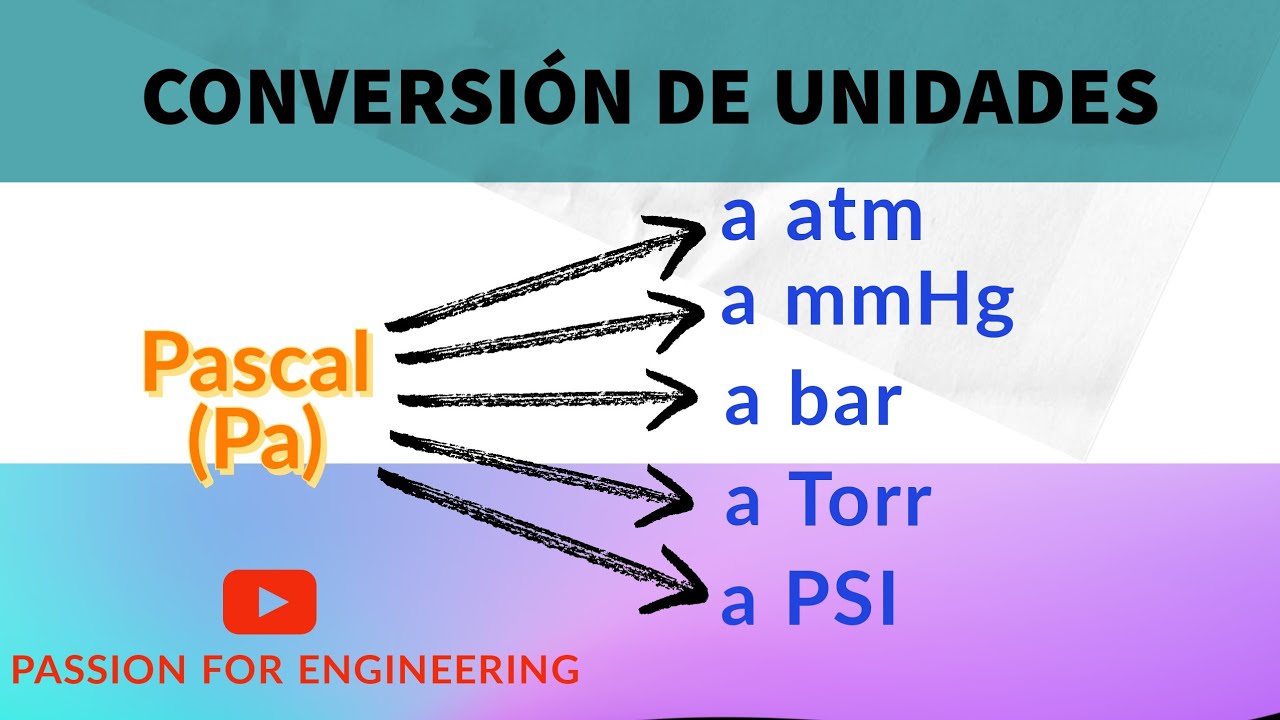
pressure, conversion units into defferent units,atm,bar,torr,psi,Pascal,mmHg, numerical,and examples - YouTube



![ANSWERED] Which of the following would represent t... - Inorganic Chemistry ANSWERED] Which of the following would represent t... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/51475127-1659105541.0026274.jpeg)