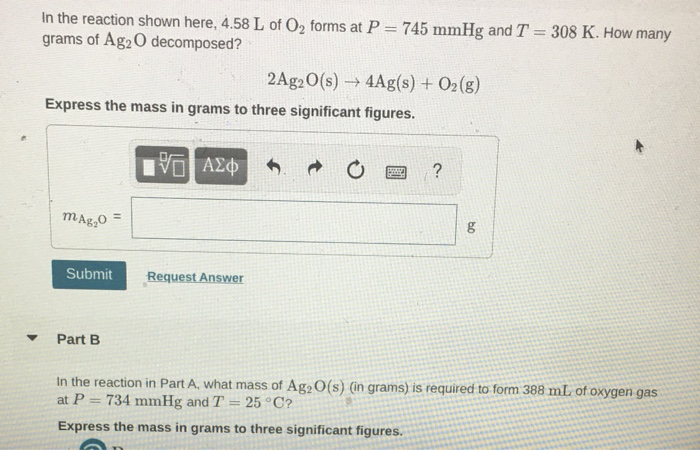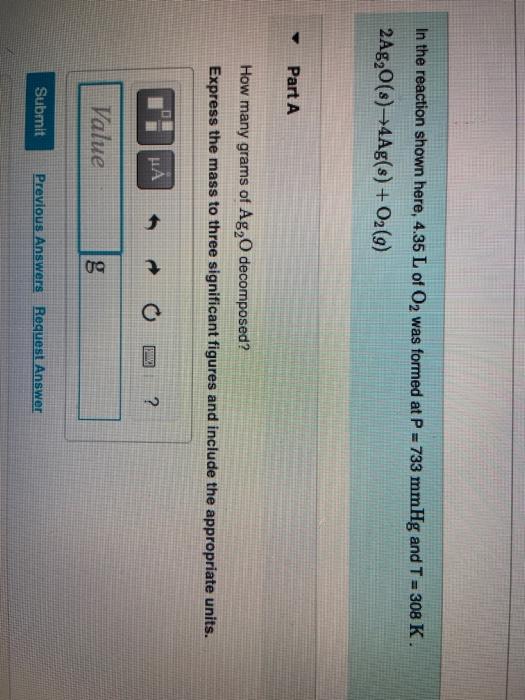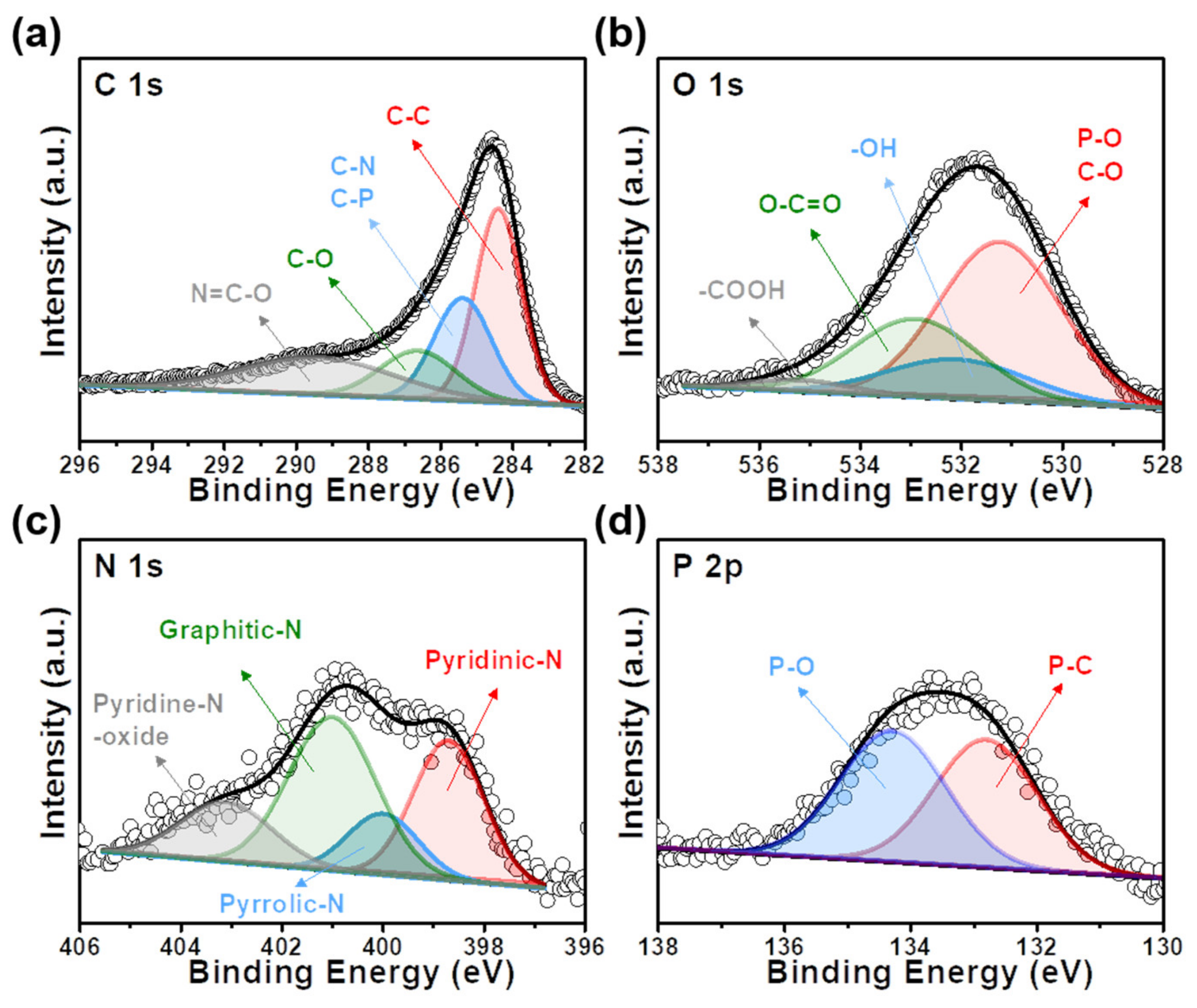
Catalysts | Free Full-Text | Accelerating the Oxygen Reduction Reaction and Oxygen Evolution Reaction Activities of N and P Co-Doped Porous Activated Carbon for Li-O2 Batteries
Equilibrium partial pressure of oxygen p(O2) for selected metal/oxide... | Download Scientific Diagram

P+O2 =P2O5 Balanced Equation|| Phosphorus plus oxygen yields Diphosphorus pentoxide Balance - YouTube

4GB 5G O2 sim only for £6 p/m, unlimited mins & texts (12 month - £72 via Moneysupermarket) @ O2 | hotukdeals

SOLVED: 1 mol of hydrocarbon P (contains C and H only) required 240 g of oxygen (O2) for complete combustion at standard state heat of combustion of P(l) is -3350 kJ/mol while

Diagram of the chemical potential of oxygen (? P O2 )-temperature (T, K) at | Download Scientific Diagram
![O - ] and R/R 0 as correlated with (K O2 P O2 ) 1/2 /L D for n = 3 or 5. | Download Scientific Diagram O - ] and R/R 0 as correlated with (K O2 P O2 ) 1/2 /L D for n = 3 or 5. | Download Scientific Diagram](https://www.researchgate.net/publication/251712737/figure/fig1/AS:298049730564096@1448072001187/O-and-R-R-0-as-correlated-with-K-O2-P-O2-1-2-L-D-for-n-3-or-5.png)